GM Seed Secrecy Threatens Organics

By Lucy Sharratt
WARNING: This seed packet could soon be full of undisclosed genetically modified (GM) seeds.
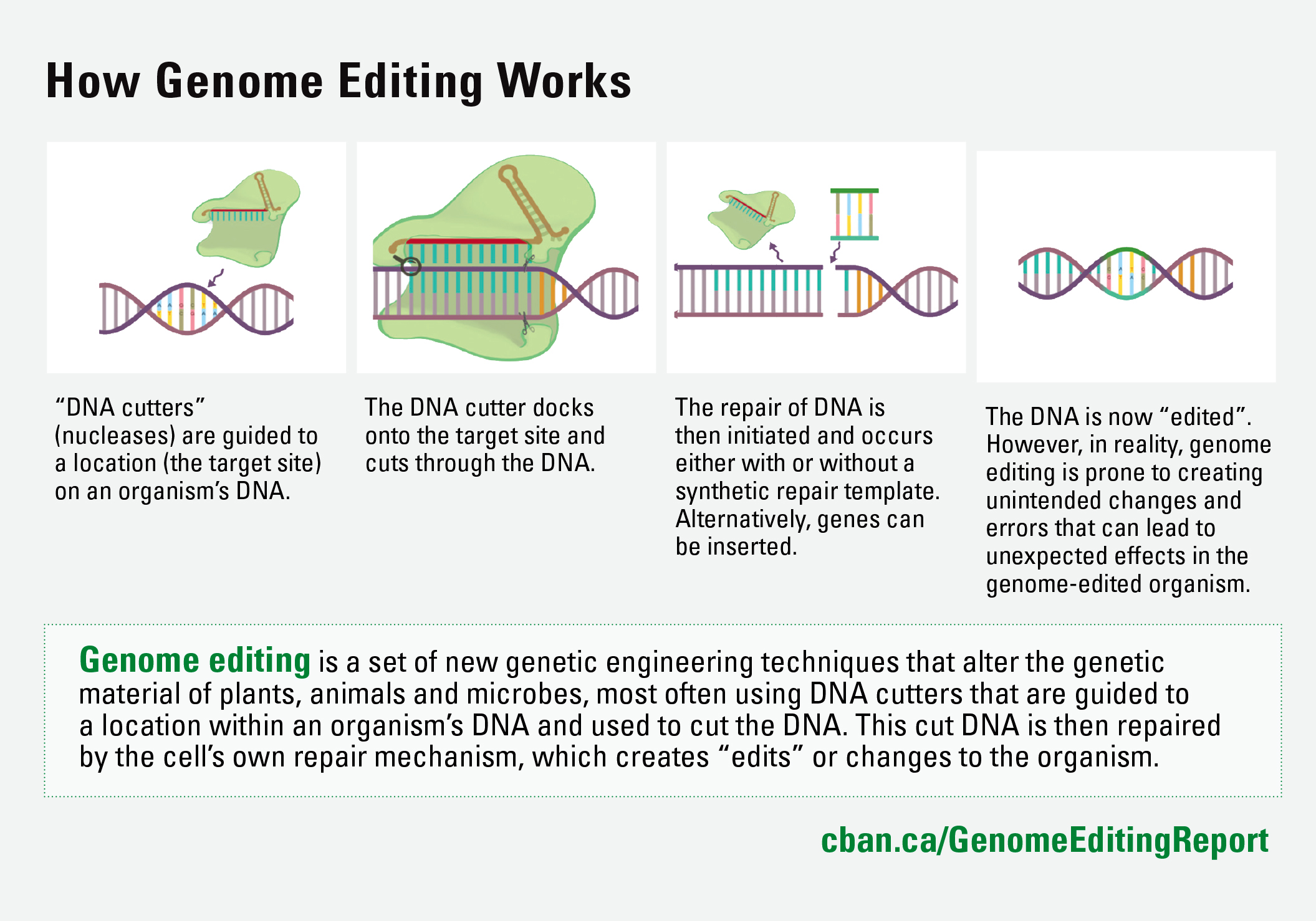
The biotechnology industry is pushing to take over the regulation of genetically engineered (genetically modified or GM) seeds and foods, and establish corporate self-regulation in place of government oversight. The Minister of Agriculture and Agri-Food is considering letting corporations conduct their own environmental safety assessments of many new gene-edited seeds with no government oversight, and decide if they want to disclose that their new product is genetically engineered. The Minister of Health has already decided that many gene edited foods don’t need any government safety checks. The proliferation of unknown genetically modified organisms (GMOs) in the food system is a serious threat to the future of organic farming.
Seed Packet Warning Sent to the Minister
At the end of last year, hundreds of farmers in the Prairies sent seed packets in the mail to the Minister of Agriculture and Agri-Food. These envelopes were empty of seeds but carried an important message to the Minister about the threat to organic farming if she allows the sale of unassessed, undisclosed genetically engineered seeds.
The Canadian Food Inspection Agency (CFIA) is proposing to allow companies to sell many new gene-edited GM seeds—those that have no foreign DNA—without notifying farmers, consumers, or even the federal government. This exemption will mean that companies will do their own environmental assessments and leave farmers dependent on companies to find out if seeds are gene-edited.
The biotechnology industry has launched a public relations campaign to characterize gene editing techniques as “plant breeding innovation” or “precision breeding,” and argue that gene-edited seeds should not even be defined as GMOs. However, gene editing is genetic engineering. This is the science, and it is also the definition in the Canada Organic Standards.
The Canada Organic Regime is overseen by the CFIA and includes the enforcement of the organic standards, which prohibit the use of genetic engineering. The Canada Organic Trade Association says, “It is the CFIA’s responsibility to ensure the environmental safety of all genetically engineered plants and the protection of the organic integrity. CFIA must continue to regulate all genetically engineered plants under the Seeds Act—including those produced through the new techniques of gene editing.”
CropLife Influence Exposed
A 2022 investigation by French-language CBC found that a CFIA document summarizing the agency’s proposals to remove regulation was actually authored by the biotechnology and pesticide industry lobby group CropLife Canada. The history of the Word file shows that CropLife’s executive director was the creator of the document.
This revelation led the National Farmers Union, CBAN, and 13 other groups to call for the CFIA President to be replaced. The groups said, “It appears that key aspects of the proposed guidance…were requested by self-interested industry groups and have been incorporated into the CFIA regulatory guidance.”
The CFIA has explained that CropLife authorship of the document was a technical mistake in the process of exchanging draft documents for comment. However, this mistake was only possible because the CFIA sent CropLife this proposal summary for their input. Farm groups such as the National Farmers Union were not asked to review it.
The CFIA consulted with farmer organizations in the summers of 2021 and 2022. However, as Garry Johnson, President of SaskOrganics said, “If the government is sincere about hearing from farmers, when their proposals would have such a major impact on organic farmers in particular, they should choose a time that is much less demanding on all of us.”
Minister Pledges GMO Transparency
In response to the media story, the Minister of Agriculture told organic farmers that they don’t need to worry; that she will make sure farmers have transparency about new GM seeds coming to the market. However, the Minister has not yet secured mandatory reporting and the CFIA continues to meet with CropLife to discuss the industry’s proposal for “voluntary transparency.”CropLife is pushing to be the arbiter of information for farmers. It wants to manage a public list that relies on biotech companies to voluntarily disclose their GM seeds. In fact, CropLife has its own private, voluntary product notification system ready to roll. This is a tactic to avoid regulation and mandatory reporting.
The biotech industry wants the Minister to accept voluntary corporate notification as sufficient transparency. However, there would be no way to know that this list was complete or to verify the information on the it.
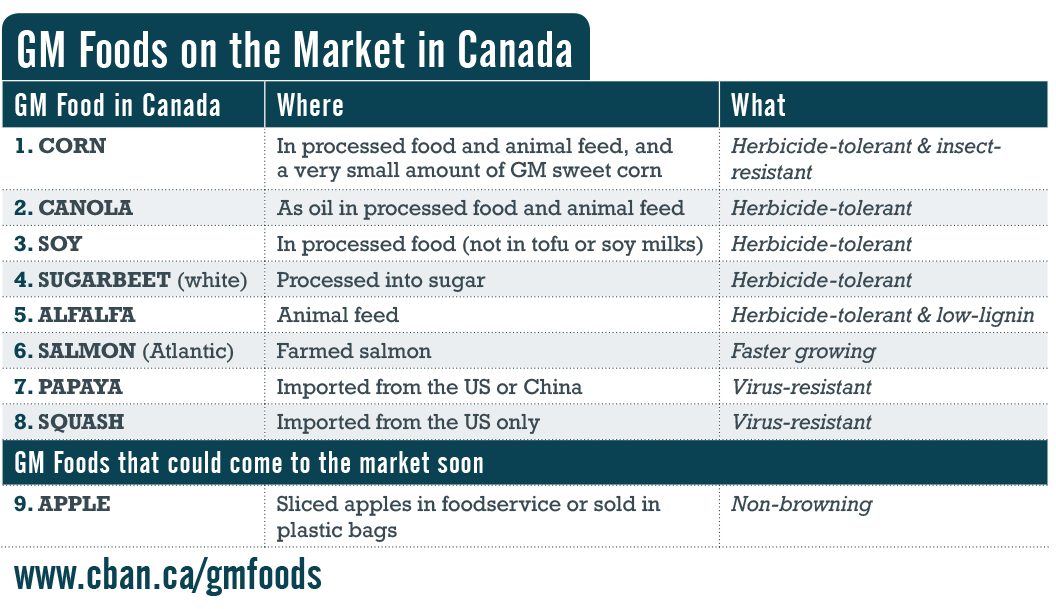
This lack of transparency would go far beyond the existing problem of unlabelled GM foods in our grocery stores. Some GM foods and seeds would be sold, planted, and eaten without the public, farmers, and government knowing that they even exist.
Removing regulation would remove the ability of the federal government to request information from companies about their new unregulated GM foods and seeds. There would be no way to track and trace these secret, unregulated GMOs.
In an October 2022 letter to the Minister, SaskOrganics, the National Farmers Union, and the Canadian Biotechnology Action Network (CBAN) argued that “transparency and traceability will only be possible if the CFIA and Health Canada retain their regulatory authority over all genetically engineered products such that, prior to release into the environment or onto the market, the departments require submission of information from product developers and have the ability to provide it to Canadians in a timely, trusted, and accessible way.”
The CFIA is stuck with an irreconcilable contradiction. The CFIA, federal government, and Minister have all publicly committed to “transparency,” but the CFIA is proposing to remove the very regulation that would ensure it.
Act Now to Stop Corporate Self-Regulation
In May 2022, Health Canada announced that it will not regulate gene-edited GM foods, and will propose amendments to the Novel Food Regulations to formalize these changes. This is an acknowledgement that the Minister of Health can still reverse Health Canada’s new regulatory guidance that exempts these foods.
In a January 2023 letter responding to SaskOrganics, the Minister of Agriculture said she has asked the CFIA and Agriculture Canada “to propose options to ensure traceability and transparency of genetically engineered plant varieties to maintain the integrity of Canada’s organic food production system.” However, the solution needed can only be secured if organic farmers, organic sector groups, and the public continue to communicate with the Minister.
The Endgame is Complete Corporate Control
CropLife members include the biggest seed and pesticide companies in the world. These companies would clearly be the beneficiaries of an open door to sell unregulated GMOs. These corporations would be free to put many GM products on the market without the costs of submitting safety data to regulators, could cut their confidential safety studies down to a bare minimum, and could keep their GMOs a market secret and thus avoid the risk of consumer boycotts. The proliferation of undisclosed GMOs would also remove the competition: by contaminating the entire food system with secret GMOs, over time, organic production and other non-GMO choices would become unviable.
Actions and updates are posted at: cban.ca/NoExemptions
Lucy Sharratt is Coordinator of the Canadian Biotechnology Action Network (CBAN), a project of the MakeWay Charitable Society.
Featured image: Letters and seed packets. Credit: Canadian Biotechnology Action Network.